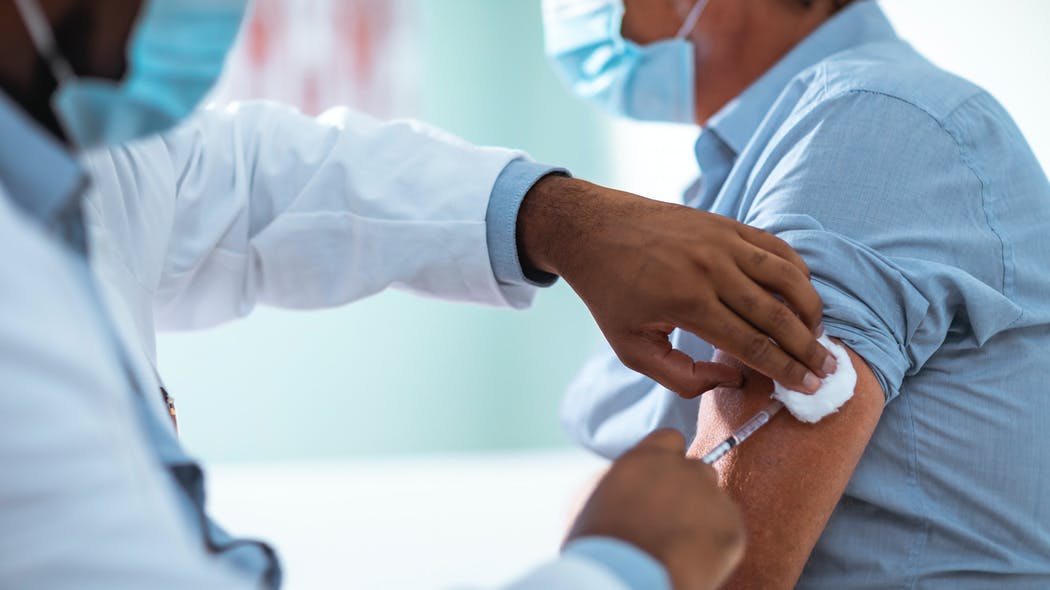
An American study has made it possible to establish an estimate in the United States of the risks of severe allergic reactions when the Pfizer / BioNTech vaccine against COVID-19 is administered. If these are present, they are rare and well taken care of.
The European Medicines Agency and then the European Commission gave the green light on December 21 to Pfizer and BioNTech’s vaccine against Covid-19 . This messenger RNA vaccine called Comirnaty (BNT162b2) is therefore the first vaccine to have obtained conditional marketing authorization in Europe: in France, it can be used there to vaccinate adults to prevent Covid 19 due to the virus. SARS CoV 2. But the technology on which this new type of vaccine is basedso-called “nucleide acid vaccines” (the vaccine from the Moderna laboratory also uses this technology) has raised many questions in the population.
Regarding possible allergic reactions, a study published by the Centers for Disease Control and Prevention in the United States is reassuring on this subject. She claims that about one in 100,000 people who have received Pfizer-BioNTech ‘s vaccine have had severe allergic reactions , noting that the benefits of vaccination far outweigh the known risks. To reach this conclusion, the researchers documented 21 cases of anaphylactic shock, the most severe manifestation of allergy, after the administration of 1,893,360 injections from December 14 to 23. All 21 cases were between 27 and 60 years old (median age 40) and all but two were treated with epinephrine.
COVID-19 infection more dangerous than allergies
In addition, 19 of the cases occurred in women , and the median time to onset of symptoms was 13 minutes, but ranged from two to 150 minutes. Four patients were hospitalized, three of them in intensive care, and the other 17 were treated in an emergency department. All but one of the patients were recovered and discharged home at the time of writing, except one, and no deaths are reported. The symptoms listed included a rash, feeling of suffocation (closing of the throat), a swollen tongue, hives, difficulty breathing , hoarseness, swollen lips, nausea and a persistent dry cough.
“On average, this represents a rate of 11.1 anaphylactic shock per million doses administered,” Nancy Messonnier, a CDC official, told reporters. The organization recalls that in comparison, influenza vaccines causeapproximately 1.3 cases of anaphylaxis per million doses administered, so the rate of anaphylaxis for Pfizer vaccine is approximately ten times higher. But still, cases of anaphylaxis linked to the Pfizer / BioNTech vaccine “are exceedingly rare and it remains in the interest of people to take the vaccine, especially in the context of the COVID-19 pandemic which represents a danger much greater for their health. ” adds Nancy Messonnier.
People at risk informed in advance
Because this type of allergic reaction is well known. “We know how to treat anaphylaxis and have arrangements in place to ensure that at vaccination sites, people who administer the vaccine are ready to treat anaphylaxis,” she says. The United States has so far authorized two vaccines: by Pfizer and by Moderna, which require two doses a few days apart. Health officials have issued the same warnings: People with a history of allergic reactions to ingredients in vaccines should avoid them. And those who have had a severe reaction to the first injection are advised not to take the second injection.
Investigations are underway to determine what could be the cause of these allergies, knowing that there is not yet enough data to know what is the rate of anaphylaxis for the Moderna vaccine or if a difference between the two vaccines. will appear. A preliminary hypothesis for these allergic reactions is the presence of a compound, polyethylene glycol (PEG). The latter has never been used before in a vaccine but is found in several everyday products: laxatives, shampoos and toothpastes. Both vaccines use PEG molecules as part of the protective shell around their main ingredient, mRNA, which transmits genetic instructions to cells.